FEops
a materialise company
At FEops, we envision a future in which cutting-edge, AI-based technology helps physicians alter the course of heart disease. Our cloud-based structural heart procedure planning platform, FEops HEARTguide™, analyses cardiac computed tomography (CT) scans and offers unprecedented insight into a patient’s total heart health.
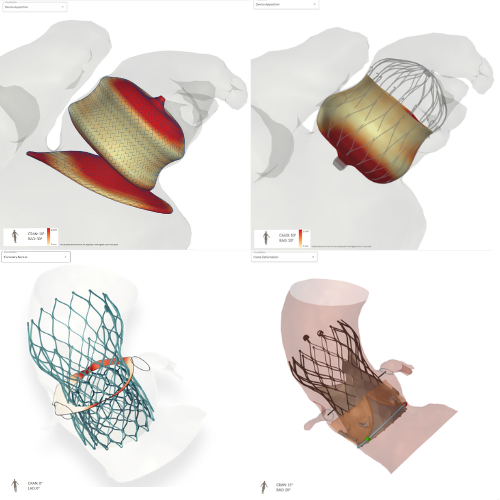
New version of FEops HEARTguide™ with workflow for TAVI Lifetime management*
11461
Patients
38
Countries
466
Hospitals
Discover Our Leading-edge, Cloud-based Structural Heart Procedure Planning Platform
FEops HEARTguide™ combines predictive simulation technology and artificial intelligence to deliver AI-based anatomical measurements and previously inaccessible data about a patient’s heart health. These insights improve procedure efficiency and outcomes* by enabling physicians to more accurately predict how transcatheter structural heart devices will interact with a specific patient’s anatomy.
Our Solutions
Patient care for structural heart disease*
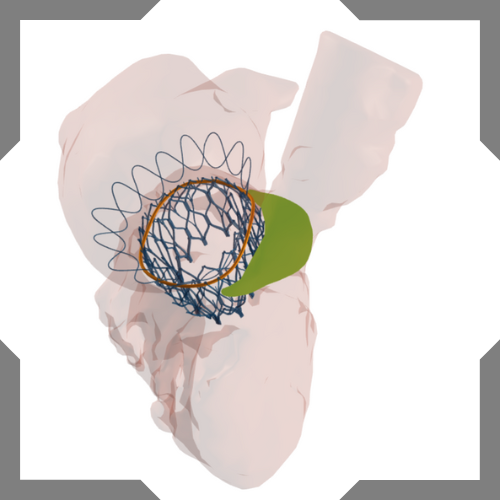
Left atrial appendage occlusion
Get unique insights to make your LAAO procedures more predictable.
Transcatheter aortic valve Implantation
Plan even your most challenging TAVI cases with confidence.
Understand options for future valve-in-valve procedures.
More exciting innovations in the FEops pipeline
Transcatheter mitral valve replacement (TMVR) ***
Early evidence supports the use of FEops simulation technology in TMVR.
Services for cardiovascular device companies
Streamlining and accelerating research and development
Finite Element Analysis (FEA) offers a highly efficient way to explore design options of cardiovascular devices.
Our Community
*In EU, UK, Canada and Australia, FEops HEARTguide™ is available for AI-enabled anatomical analysis and computer simulation with indication for use in TAVI and LAAO. In USA, FEops HEARTguide ™ is available for AI-enabled anatomical analysis with indication for use in TAVI and LAAO and for computer simulation in LAAo with WATCHMAN™, WATCHMAN™FLX, Amplatzer™ Amulet™. New TAVI Lifetime Management is not available for clinical use in the US, 510(k) pending.
**Hokken T. et al. Clinical value of CT‐derived simulations of transcatheter‐aortic‐valve‐implantation in challenging anatomies the PRECISE‐TAVI trial. CCI Catheterization and Cardiovascular Interventions, Sept. 2023 -DOI: 10.1002/ccd.30816.
***FEops HEARTguide is currently not indicated for clinical use in TMVR